The applied deconvolution strategy may provide a simple guideline to obtaining high-quality fits to experimental data on the basis of a careful evaluation of experimental conditions, sample properties, and the limits of the fit procedure. The sp2 peak, characteristic of aromatic carbon, features a strong asymmetry that changes with the curvature of the sample surface and, thus, cannot be neglected in spectral analysis. These findings illustrate that both spectral line shapes and binding energy components must be considered in the analysis of potentially defective surfaces of carbon materials. Some of the steps employed for the static iterative Shirley background removal are shown. However, if one additionally allows a variable displacement of the sigmoidal step relative to the peak, excellent fitting results can be obtained. a) Au 4p3/2 XPS spectrum from a clean gold surface. With high-resolution XPS and XPS depth profiling, the spectral components arising from disordered carbon and surface-defect states can be distinguished from aromatic sp2 carbon. While this approach has been successfully applied to vibrational spectra, we find that the resulting curves are less suitable for least-squares fits of X-ray photoelectron spectroscopy (XPS) data. A new method for the fitting of X-ray photoelectron spectra using an advanced Tougaard-background model for laterally inhomogeneous samples is presented. 10 ), enabled adequate fitting of the experimental spectrum ( Fig.
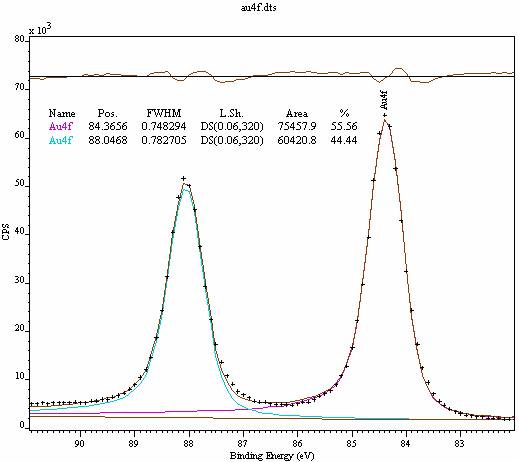
The addition of a third, low intensity peak O3, to account for CO bonds related to the detected oxidised adventitious carbon (see Fig. Controlled manipulation of such samples is performed by annealing, sputtering, and oxygen functionalization to identify different CC bonding states and assess the impact of the manipulations on spectral line shapes and their binding energy positions. Peak fitting the obtained spectrum using two components, with an O1:O2 peak area ration of 2:1 did not lead to satisfactory results. In this work, an overview of extrinsic and intrinsic effects that influence the C 1s XPS spectra-for example, photon broadening or carbon–catalyst interaction-of various graphitic samples is presented. Then: Peak-fit each Chemical State Spectra.

Next: If sample was Non-Conductive, then correct all spectra by using Charge Referencing method. As a result we can not see the different Chemical State signals until we change the instrument settings to give high energy resolution settings.

The SurveyID provides automatic XPS peak identification and quantification. Survey Spectra use low energy resolution settings.
#Xps peak fitting procedures software
However, the analysis of XPS data-in particular the C 1s region-can be complex, impeding a straightforward evaluation of the data. Avantage is the Software package supplied with Surface Analysis Instruments. X-ray photoelectron spectroscopy (XPS) is a widely used technique for characterizing the chemical and electronic properties of highly ordered carbon nanostructures, such as carbon nanotubes and graphene.
